High Homocysteine: The Genetic Root Cause for Chronic Inflammation
Are you dealing with unexplained chronic pain, fatigue, brain fog, or "undiagnosable" inflammation? If standard lab tests come back "normal" but you feel like your body is on fire, high homocysteine levels could be the silent culprit. As a global genetic root cause consultant, Dan Purser MD has treated thousands of patients worldwide—from the US East Coast to Dubai, Norway, and the UK—who've struggled with these symptoms. Is there a treatment for genetic high homocysteine inflammation? Homocysteine isn't just a lab marker; it's a biochemical alarm signaling deeper genetic and metabolic breakdowns that drive inflammation, oxidative stress, and serious health risks. In this guide, we'll break down what homocysteine is, its pitfalls, the "nasty" genes behind it, and our proven protocol to address it at the root.
What Is Homocysteine and Why Does It Matter?
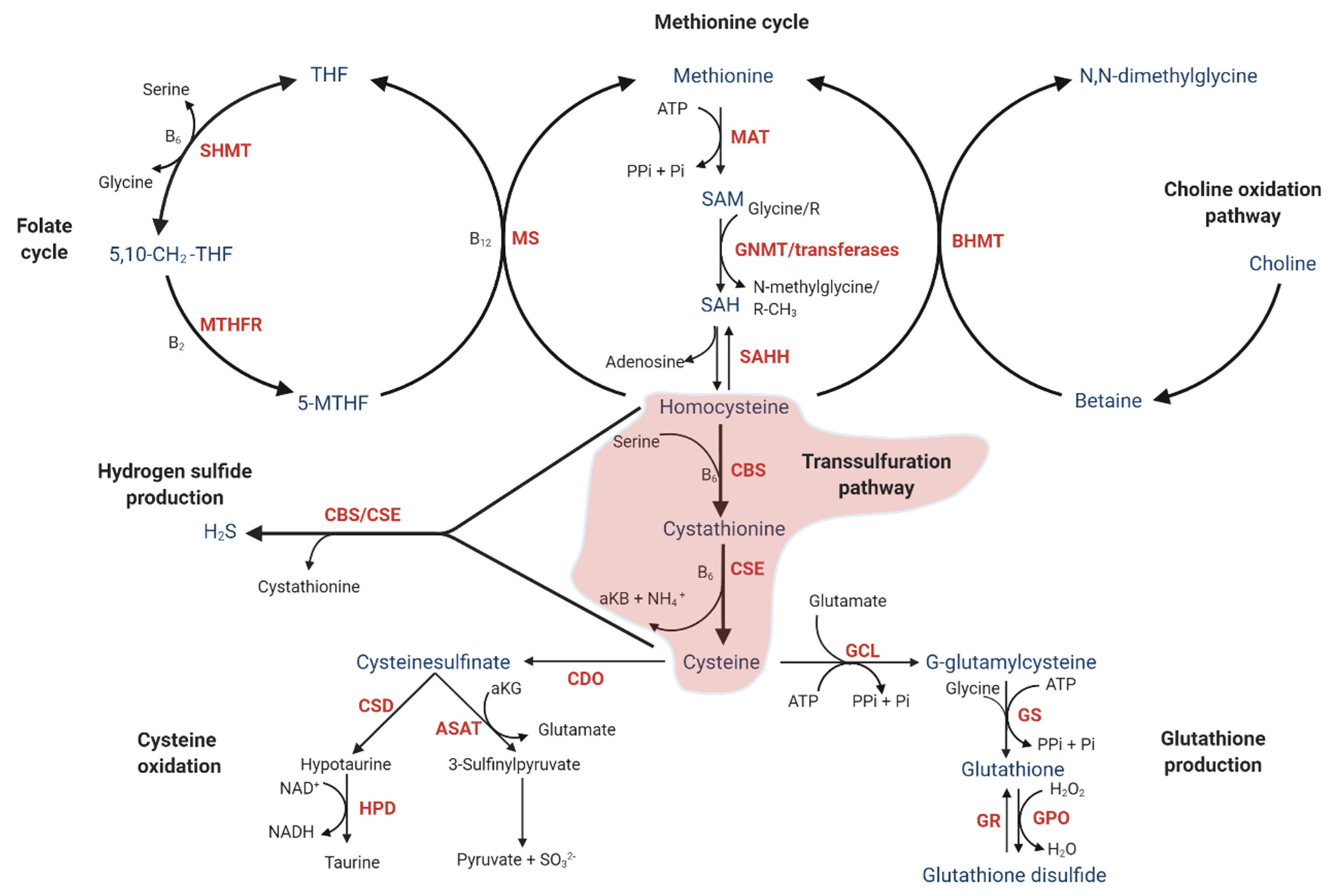
Homocysteine is a naturally occurring amino acid produced in every cell during methylation - a critical process for DNA repair, hormone balance, neurotransmitter production, and detoxification. In healthy systems, homocysteine is quickly recycled back into methionine (via methylation) or converted into cysteine and glutathione (via the transsulfuration pathway). Glutathione, your body's master antioxidant, then neutralizes oxidative stress, quenches free radicals, and clears toxins like heavy metals.
But when this pathway stalls - often due to genetics - homocysteine builds up. Elevated levels (hyperhomocysteinemia) act like a "trigger," stimulating immune cells (monocytes) to release pro-inflammatory cytokines such as IL-8 and MCP-1. This creates a cascade of systemic inflammation, oxidative damage, and detox failure. Normal homocysteine levels should be under 10 μmol/L, but genetic factors can mask issues in standard blood tests, especially if you're malnourished in key B vitamins.
From Dr. Purser’s insights (e.g., "Homocysteine: The Hidden Marker Behind Pain, Fatigue, and Inflammation"), high homocysteine isn't random - it's almost always genetic, involving disruptions in 17+ key steps of the transsulfuration pathway. This "traffic jam" leads to glutathione shortages, making your body vulnerable to everyday stressors like infections, toxins, or even stress itself.
Elevated Cytokines: A Silent Threat to Your Health?
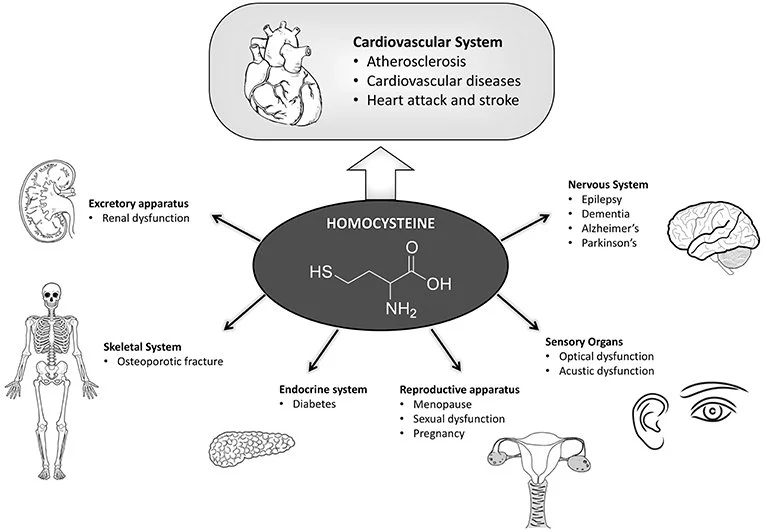
High homocysteine doesn't cause direct damage; it amplifies inflammation and oxidative stress, setting the stage for chronic conditions. Based on clinical patterns and research shared in my articles like "High Homocysteine, MTHFR Mutations, and the Inflammation Crisis," here are the major risks:
Chronic Inflammation and Pain: Elevated homocysteine ramps up cytokines like IL-8 (neutrophil recruiter) and MCP-1 (monocyte attractor), leading to widespread aches, muscle tenderness, and "thermal runaway" inflammation. This is a core driver in fibromyalgia, where detox failure heightens tissue sensitivity.
Cardiovascular Dangers: It promotes endothelial dysfunction, arterial stiffness, blood clots, and vascular injury. Linked to "invisible" heart attacks, strokes, hypertension, and chronic kidney disease (CKD). In Dr. Purser’s post "Can Glutathione Reverse Kidney Disease," he explains how homocysteine's oxidative effects can progress CKD stages, but targeted support may improve creatinine trends and renal function.
Fertility and Hormonal Issues: Impairs ovarian function and hormone metabolism, contributing to PCOS (with insulin resistance and infertility), recurrent pregnancy loss, and hormonal imbalances. High levels exacerbate cytokine-driven inflammation in reproductive tissues.
Neurological and Mental Health Risks: Causes brain fog, anxiety, depression, cognitive decline, and even increased stroke risk. It disrupts neurotransmitter balance and methylation, worsening mood instability.
Kidney and Detox Failures: Slows heavy metal clearance (e.g., copper, mercury), leading to toxin buildup, kidney fibrosis, and potential failure. Also tied to sarcoidosis, psoriasis, Crohn's disease, and autoimmune persistence.
Other Long-Term Risks: Accelerated aging, reduced neurogenesis, diabetes, neuropathy, and higher cancer susceptibility due to DNA repair issues and chronic oxidative stress.
“Underlying genetic SNPs causing cytokine overload is the ROOT of inflammation in the body. ”
The "Nasty" Genes Fueling Homocysteine Chaos
From Dr. Purser’s teachings "Unlocking the Root Cause of Inflammation," homocysteine issues stem from single nucleotide polymorphisms (SNPs) in genes controlling methylation, transsulfuration, and cytokine regulation. These "arsonists" and "anchors" create a state of "redox bankruptcy," where your body can't produce enough glutathione, SOD, or catalase to fight back.
We test for these in our DRP Genetics panels (detailed below), drawing from comprehensive data like our cytokine genetics database. Here's a table of the top 10 most problematic homozygous SNPs, their triggered cytokines/markers, and associated diseases/symptoms (based on clinical observations and genetic data):
| Rank | Genetic SNP (Homozygous) | Primary Cytokine/Marker Triggered | Main Disease or Symptom |
|---|---|---|---|
| 1 | IL-6 (-174 G/G) | Interleukin-6 (IL-6) | The Genetic Arsonist: Causes "thermal runaway" inflammation, deep tissue fire, and systemic exhaustion. |
| 2 | IL-2R (sIL-2R) | Soluble IL-2 Receptor | The Anchor: Leading cause of Sarcoidosis and "Immune Amnesia" (failure to produce IgG). |
| 3 | IFN-γ (+874 T/T) | Interferon-gamma | The Viral Ghost: Drives Macrophage Madness and the formation of granuloma "fortresses" in Sarcoidosis. |
| 4 | CBS (C699T / 844ins68) | Homocysteine / IL-8 | The Broken Filter: Disrupts the Transsulfuration Pathway, leading to Chronic Kidney Disease (CKD) and hypertension. |
| 5 | IL-1β | Interleukin-1 beta | The Match: Triggers the NLRP3 inflammasome, causing deep bone pain, rigors, and high-spike fevers. |
| 6 | TGF-β | Transforming Growth Factor Beta | The Scar Maker: Drives systemic Fibrosis, turning soft organ tissue into a "fossilized" state. |
| 7 | MCP-1 (-2518 A/A) | Monocyte Chemoattractant Protein-1 | The Recruiter: Breaches the blood-brain barrier to cause Cognitive "Brain Fog" and monocyte infiltration. |
| 8 | MTHFR (C677T) | Homocysteine / IL-8 | The Attack Dog: Acts as a proxy for high cytokine sensitivity, driving Strokes, DVT, and Heart Attacks. |
| 9 | IL-10 (-1082 A/A) | Interleukin-10 (Deficiency) | The Broken Brake: Genetic inability to stop a flare, leading to Perpetual Inflammation that never resolves. |
| 10 | IL-17 / IL-23R | Interleukin-17 & 23 | The Barrier Breach: Triggers a "civil war" on body surfaces, manifesting as Crohn’s Disease or Psoriasis. |
Table: Top 10 Genetic "Arsonists" and "Anchors" from the Purser Model – Homozygous SNPs Driving Undiagnosable Inflammation
These SNPs (e.g., from MTHFR, CBS, IL6) are common culprits, often homozygous in our patients. For a full list, our panels cover 30+ cytokines influenced by homocysteine (e.g., IL-1β, TNF-α, IFN-γ) and 100+ genes/rsIDs from methylation to redox pathways.
Genetic Predisposition for Cytokine Storm
Cytokine storm - also known as cytokine release syndrome or hypercytokinemia - represents a life-threatening over activation of the immune system where pro-inflammatory cytokines flood the body, leading to widespread inflammation, tissue damage, organ failure, and potentially death. This phenomenon has gained massive attention in contexts like severe COVID-19, sepsis, influenza, and certain autoimmune flares. While environmental triggers (e.g., viral load, infections) play a major role, genetic predisposition often determines who escalates to a full cytokine storm versus those who mount a controlled response.
In our practice, we've observed that many patients with "undiagnosable" chronic inflammation or recurrent severe flares carry homozygous variants in key cytokine-regulating genes. These genetic "arsonists" lower the threshold for uncontrolled cytokine production, amplifying signals from stressors like high homocysteine, infections, or oxidative stress. Our DRP Genetics Panel and DRP Cytokine Genetics specifically screen for these predispositions, revealing why some individuals experience perpetual or explosive inflammation while others do not.
Key Genetic Factors Driving Cytokine Storm Risk
Genetic variants - particularly single nucleotide polymorphisms (SNPs) in promoter regions - can increase baseline or inducible production of pro-inflammatory cytokines. Common culprits include:
IL-6 (Interleukin-6) - This is often called the "master switch" for sudden, strong inflammation. A common gene change (SNP rs1800795, also known as -174 G>C) affects how much IL-6 your body makes. People with two copies of the G version (homozygous GG) tend to pump out higher levels of IL-6, which raises the risk of severe inflammation and worse outcomes in infections or the flu. The C version can sometimes act like a brake, helping moderate the response and offering a bit of protection in some groups.
TNF-α (Tumor Necrosis Factor-alpha) - This cytokine acts like a demolition crew - it tears down tissue and ramps up other inflammatory signals. A key gene variant (SNP rs1800629, often the A version) leads to higher TNF-α production. People with this version are more likely to have intense inflammation during viral illnesses or sepsis, making the storm more severe and harder to control.
IL-1β (Interleukin-1 beta) - Think of this as the spark that lights the inflammasome (a built-in alarm system in cells). It triggers high fevers, deep pain, and widespread body-wide inflammation. Common gene changes in the IL1B gene (like rs16944 and rs1143634) make your body release extra IL-1β, which is linked to stronger inflammatory reactions and more severe disease patterns in things like infections or autoimmune conditions.
Other Important Contributors:
IFN-γ (Interferon-gamma) - A variant like +874 T/T can cause macrophages (your body's cleanup cells) to go overboard, leading to excessive activation and the formation of granulomas (clumps of immune cells that wall off threats but can cause damage in diseases like sarcoidosis or tuberculosis).
IL-17/IL-23 pathway - These cytokines help defend skin and gut barriers, but genetic glitches here can cause "friendly fire" attacks on your own tissues, leading to flare-ups in conditions like Crohn's disease or psoriasis.
IL-10 deficiencies - IL-10 is the body's main "calm down" signal—it puts the brakes on inflammation. A common variant (like rs1800896) can lower IL-10 production, meaning the immune system struggles to shut off the storm once it starts, letting inflammation run wild and harder to resolve.
These variants often interact with high homocysteine (which upregulates IL-8, MCP-1, and other chemokines via oxidative stress), creating a perfect storm in genetically susceptible individuals.
Root Cause Genetic Inflammation Protocol
Standard medicine uses "Plan A" - biologics that block signals but ignore the fire. Our "Plan B" focuses on "VARS" (Validated, Absorbable, Reduced, Stable) protocols to quench superoxides and drain the peroxides that weld your inflammatory switches in the "ON" position. By reopening your "IgG Factory" and providing the only patented oral reduced functional glutathione, we move beyond management and into systemic restoration.
Blind supplementation won't fix homocysteine and cytokines - genetics demand precision. Our “Plan B” protocol shifts from suppression to restoration, using biotech-driven tools to reopen your IgG factory and lift inflammatory anchors.
Step 1: Advanced Testing for Clarity
DRP Genetics Panel: A saliva-based test screening 17+ SNPs in homocysteine metabolism (e.g., MTHFR C677T/A1298C, CBS, MTR/MTRR). Identifies "broken filters" in transsulfuration and methylation.
DRP Cytokine Genes Panel: Analyzes genetic predispositions for cytokine storms (e.g., IL-6, IL-8, MCP-1, IFN-γ). Reveals how homocysteine amplifies inflammation via 30+ cytokines.
CMA (Comprehensive Micronutrient Analysis) with Redox: Bonus intracellular testing for nutrient deficiencies (e.g., B6, B12, folate, molybdenum) and redox status. Uncovers hidden gaps that blood tests miss, like SOD/Catalase shortages.
These panels provide a "source code" view, explaining why you're inflamed despite normal labs. Available remotely worldwide via Zoom consultations.
Step 2: Targeted Treatment with the VARS® Trio Protocol
Once tested, we deploy the VARS Trio - the ultimate support for genetic-rooted inflammation caused by homocysteine genes and cytokines. VARS stands for Validated, Absorbable, Reduced, Stable biologicals, using patented forms for maximum efficacy:
VARS Glutathione: The only oral reduced functional glutathione that lowers cytokines (e.g., IL-8, MCP-1), quenches oxidative stress, and restores detox. As per my article "The Role of Reduced Glutathione in Modulating Cytokine Release," it silences inflammatory signaling in 72-90 days.
VARS SOD (Superoxide Dismutase): Neutralizes superoxide radicals, preventing "molecular glue" that welds inflammatory switches "ON." Essential for mitochondrial protection and cancer prevention.
VARS Catalase Support: Acts as a "metabolic drain" for hydrogen peroxide, allowing tissue repair and reducing chronic oxidative stress.
This trio overrides genetic switches, supports the "redox floor," and synergizes with your existing treatments (don't stop "Plan A" without your doctor's input). Patients often see a cytokine normalization, and symptom relief - backed by before-and-after labs.

Ready to End Your Undiagnosable Inflammatory Nightmares?
High homocysteine and overblown cytokines aren’t a life sentence - they’re a solvable genetic puzzle. If you're tired of masking symptoms and ready for root cause resolution, book a global Zoom consultation today. As your genetic counselor, my team will guide you through testing and a personalized VARS Trio plan.
Disclaimer: This is educational content. Consult your local physician for medical advice. Results vary based on individual genetics and compliance.
This custom-made genetic test, designed by Dr. Purser, dives deep into the key genetic factors he believes contribute to the biggest health challenges. With The Works!, you’ll gain powerful insights into your nutrition, fitness, and overall physiology, all in one comprehensive analysis.
Your personalized report will cover:
✔️ Methylation & DNA Repair – MTHFR and other genetic markers influencing wellness
✔️ Stress & Resilience – Fight-or-flight response and how your body handles stress
✔️ Detox & Antioxidants – Glutathione levels, toxin elimination, and free radical defense
✔️ Inflammation & Recovery – How your body reacts to inflammation and repairs cells
✔️ Brain Chemistry & Energy – Neurotransmitter balance, overall energy production, and metabolism
✔️ Weight, Nutrition & Cardiovascular Health – Blood sugar levels, vitamin deficiencies, fat metabolism, and exercise response
What’s Included
🩺 30-Minute Consultation with Dr. Purser – Get expert insights and a breakdown of your results
💊 Custom Supplement Guide – Receive tailored recommendations with direct links to purchase
Take charge of your health with data-driven guidance! Order now for personalized, science-backed wellness strategies.
YOUR FAST‑TRACK TO DEEP, CELLULAR‑LEVEL HEALTH
Unlock the test Dr. Purser trusts most for identifying the hidden nutrient deficiencies that hold your metabolism, energy, and long‑term wellness back. This exclusive bundle includes a one‑on‑one consultation with Dr. Purser, giving you expert interpretation and a personalized plan based on your results.
The CMA goes far beyond standard bloodwork. Instead of measuring nutrients floating in your bloodstream, it analyzes what’s happening inside your immune cells—where real metabolic activity takes place. This means you get a true, long‑term picture of your nutrient status, oxidative stress, and cellular resilience.
WHY THE CMA MATTERS
✔️ Identifies Hidden Deficiencies — Vitamins, minerals, amino acids, and more
✔️ Reveals Oxidative Stress Load — Understand how toxins, medications, and daily exposures impact your cells
✔️ Shows True Metabolic Function — How efficiently your cells produce energy
✔️ Provides Long‑Term Insight — Not just a moment‑in‑time snapshot
This is the test for people who want answers, clarity, and a targeted plan—not guesswork.
WHO BENEFITS MOST
Anyone wanting a personalized nutrition & supplement strategy
Women navigating fertility, pregnancy, perimenopause, or menopause
Individuals with skin issues, fatigue, mood swings, or low vitality
High performers, athletes, and those under chronic stress
People preparing for or recovering from surgery
Those managing weight, metabolic syndrome, or chronic conditions
If you’ve been told “your labs look normal” but still don’t feel your best, this test is built for you.
WHAT’S INCLUDED
🩺 30‑Minute Consultation with Dr. Purser Your results, explained clearly—with actionable next steps.
💊 Customized Supplement & Nutrition Plan Tailored recommendations based on your cellular needs, with direct links for easy ordering.
Your kit includes everything needed for your blood draw. You’ll simply schedule your draw with a qualified provider (fees vary). We typically recommend AnyLabTestNow, and you can easily find a location near you at: www.anylabtestnow.com/locations
Once your sample is collected, reach out to our office and we’ll book your results appointment with Dr. Purser.
YOUR HEALTH DESERVES MORE THAN GUESSING
Get the data, the clarity, and the expert guidance you need to finally move forward. Order your CMA bundle today and take the first step toward deeper, long‑lasting wellness.


