The Hidden Molecule Driving Inflammation: Understanding the Purser Model of Homocysteine
Most people have never heard of homocysteine, yet this small molecule may hold the key to understanding inflammation, cardiovascular disease, neurological problems, and many chronic conditions.
For decades, homocysteine was viewed as a toxic substance in the blood. Doctors were taught that lowering it with B-vitamins might reduce heart disease risk. But research results were inconsistent, leaving many physicians unsure whether homocysteine truly mattered.
New research and clinical observations are changing that view.
We introduce a new framework called the Purser Model, which explains why homocysteine may be far more important than previously believed.
Homocysteine: A Signal, Not Just a Toxin
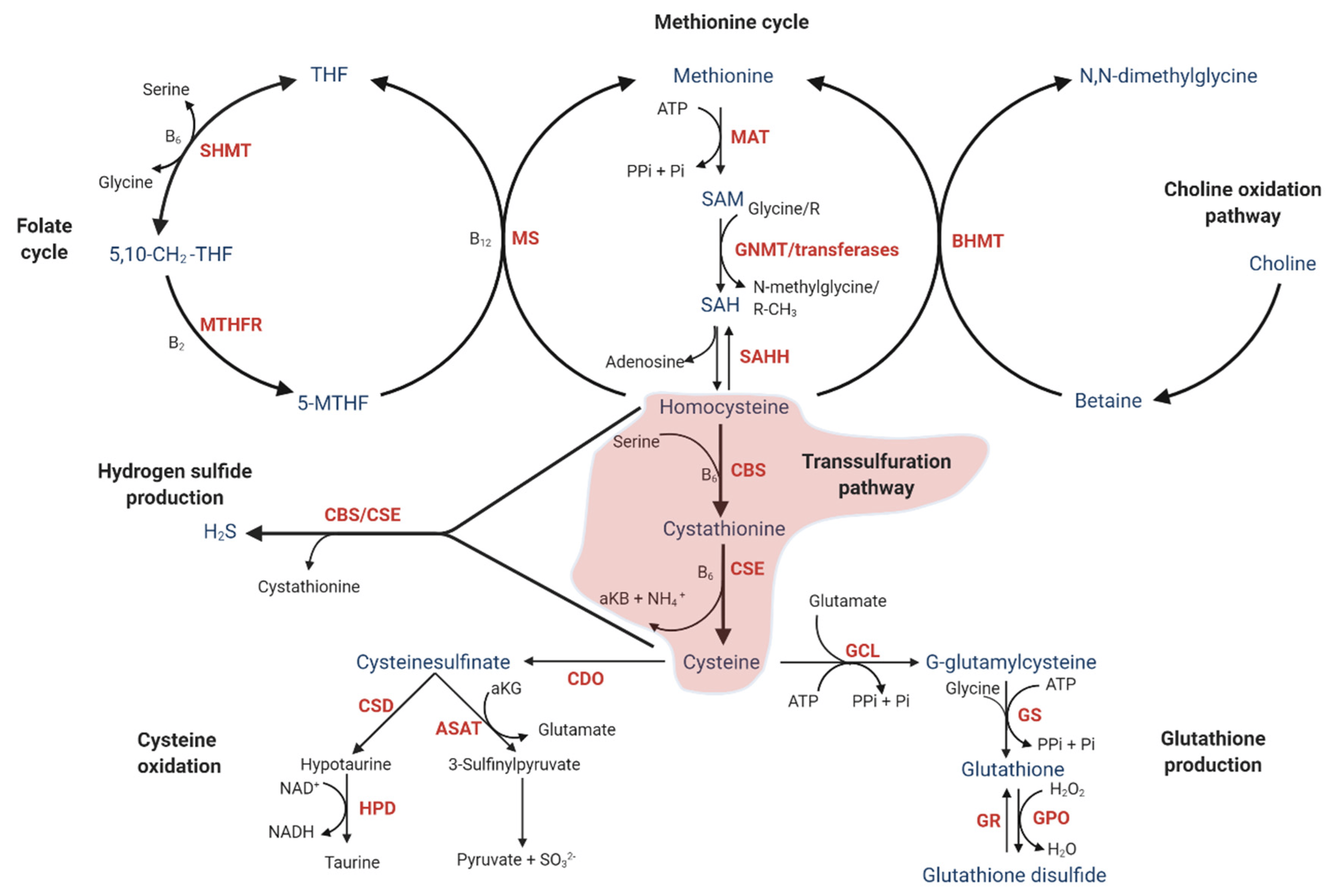
Homocysteine is a natural byproduct of protein metabolism. Your body normally recycles it through several metabolic pathways that require nutrients like:
Vitamin B6
Vitamin B12
Folate
Serine
Glutathione
When those pathways work properly, homocysteine stays in balance.
But when the system becomes disrupted—because of nutritional deficiencies, genetics, or oxidative stress—homocysteine begins to accumulate.
Traditionally, the focus was on lowering the number itself.
The Purser Model suggests something more important is happening.
The Real Problem: Cytokine Activation
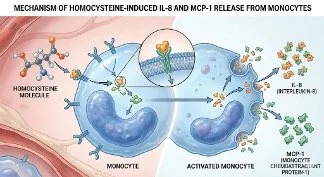
According to the Purser Model, homocysteine is not the primary cause of tissue damage. Instead, it acts as a marker of cytokine sensitivity.
Cytokines are inflammatory signaling molecules released by immune cells.
When homocysteine rises, it can trigger immune cells—especially monocytes and endothelial cells—to release inflammatory cytokines such as:
IL-8
MCP-1
These cytokines recruit immune cells and amplify inflammation throughout the body.
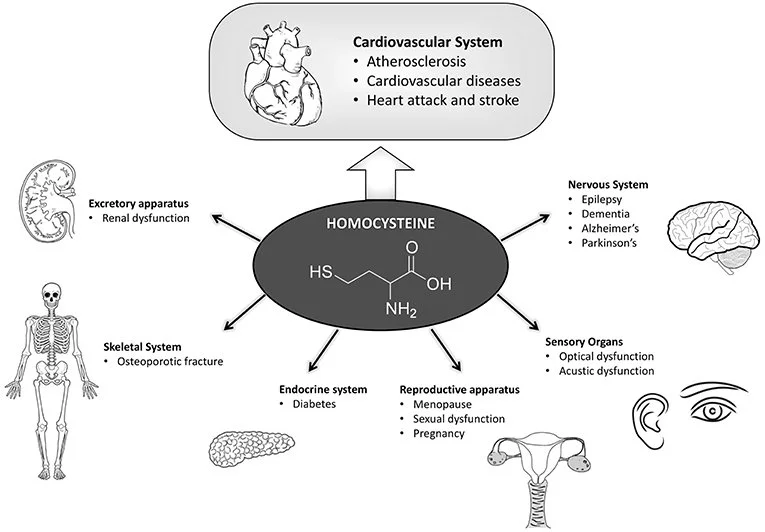
This cascade can lead to:
vascular injury
kidney inflammation
oxidative stress
blood clot formation
neurological damage
In other words, homocysteine may act like the smoke alarm, while cytokines are the fire.
Risks Associated with Elevated Homocysteine
Genetics: Why Some People Are More Vulnerable
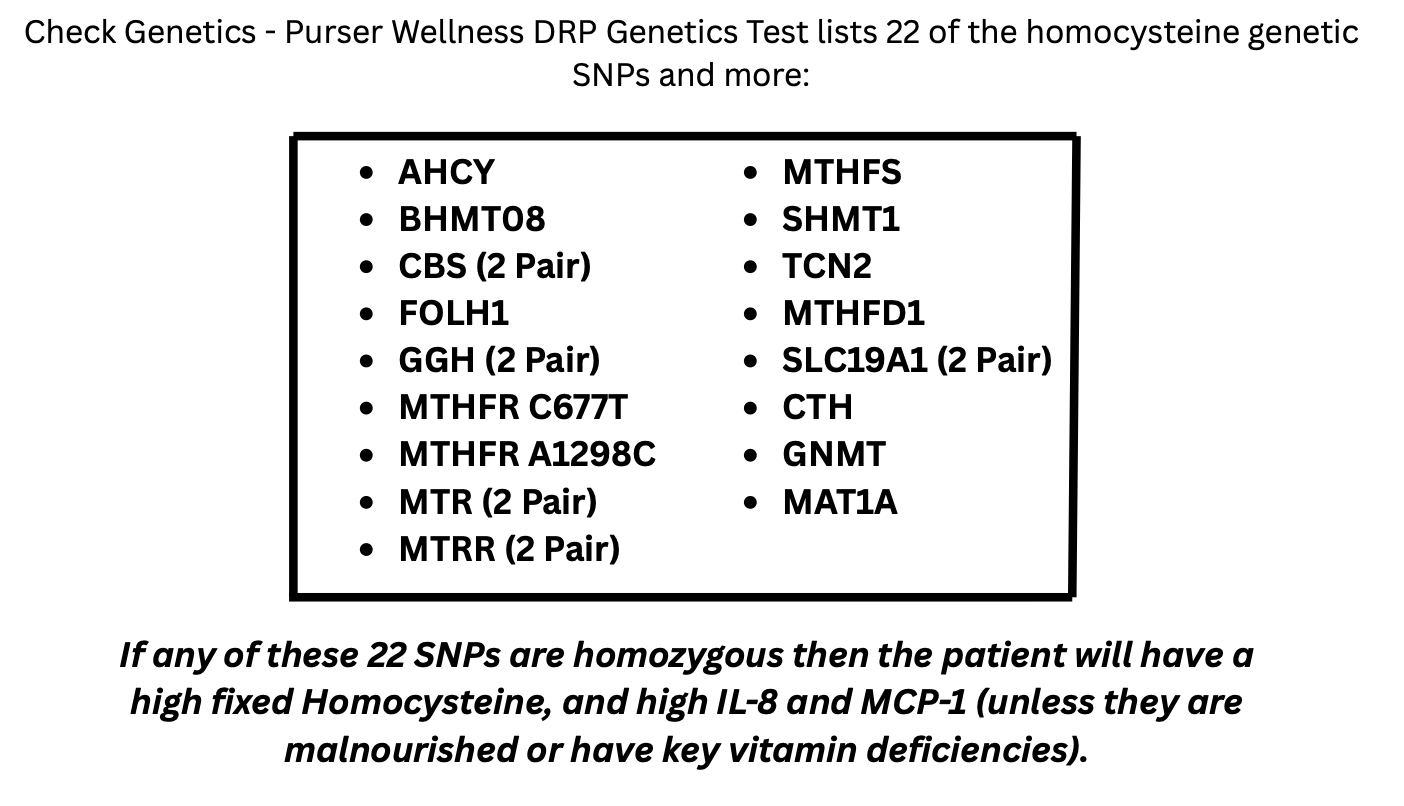
Another major discovery described in the updated book is the identification of 22 genetic variants (SNPs) that influence homocysteine metabolism.
These genes affect pathways involving:
methylation
folate metabolism
redox balance
the transsulfuration pathway
glutathione production
When certain variants occur in a homozygous state, they can significantly increase a person’s baseline homocysteine levels.
This may explain why many patients with chronic inflammation, fatigue, or unexplained illness have struggled to find answers through traditional medical testing.
Genetics sets the stage, but it is not destiny.
The Glutathione Connection
A central component of the Purser Model is glutathione, the body’s most important intracellular antioxidant.
Elevated homocysteine often correlates with reduced glutathione levels, creating a vicious cycle:
Homocysteine rises
Glutathione production falls
Oxidative stress increases
Inflammatory cytokines activate
Breaking this cycle by restoring redox balance appears to be a critical step in reducing inflammatory signaling.
A New Clinical Workflow
Instead of simply lowering homocysteine, the Purser Model proposes a more comprehensive approach.
A simplified workflow includes:
Correct nutritional deficiencies
Using intracellular micronutrient testing to restore key vitamins and amino acids.Measure homocysteine
Once nutritional status is corrected, homocysteine reflects a more accurate baseline.Evaluate genetics
Genetic testing can reveal SNP variants affecting methylation and detox pathways.Monitor inflammation risk
Cytokine activity may increase during infections, surgery, trauma, or stress.Support glutathione and redox balance
Address oxidative stress to stabilize inflammatory signaling.
This approach focuses on root cause physiology rather than isolated lab numbers.
Why This Matters
Chronic inflammation is now recognized as a driver of many major diseases, including:
cardiovascular disease
stroke
chronic kidney disease
neurodegenerative disorders
autoimmune conditions
Understanding the role of homocysteine within this broader inflammatory network may help physicians identify underlying metabolic dysfunction earlier.
More importantly, it gives patients a framework for understanding how nutrition, genetics, and cellular antioxidants interact to influence long-term health.
A Simpler Way to Understand a Complex Problem
The goal of The Purser Model is not to overwhelm readers with biochemical pathways.
Instead, it translates decades of research and clinical experience into a clear message:
Homocysteine is not just a lab value.
It is a window into how your body handles inflammation, oxidative stress, and metabolic balance.
By recognizing these underlying mechanisms, both patients and physicians can take a more informed approach to protecting long-term health.
This custom-made genetic test, designed by Dr. Purser, dives deep into the key genetic factors he believes contribute to the biggest health challenges. With The Works!, you’ll gain powerful insights into your nutrition, fitness, and overall physiology, all in one comprehensive analysis.
Your personalized report will cover:
✔️ Methylation & DNA Repair – MTHFR and other genetic markers influencing wellness
✔️ Stress & Resilience – Fight-or-flight response and how your body handles stress
✔️ Detox & Antioxidants – Glutathione levels, toxin elimination, and free radical defense
✔️ Inflammation & Recovery – How your body reacts to inflammation and repairs cells
✔️ Brain Chemistry & Energy – Neurotransmitter balance, overall energy production, and metabolism
✔️ Weight, Nutrition & Cardiovascular Health – Blood sugar levels, vitamin deficiencies, fat metabolism, and exercise response
What’s Included
🩺 30-Minute Consultation with Dr. Purser – Get expert insights and a breakdown of your results
💊 Custom Supplement Guide – Receive tailored recommendations with direct links to purchase
Take charge of your health with data-driven guidance! Order now for personalized, science-backed wellness strategies.
THE MOST COMPREHENSIVE CELLULAR HEALTH TEST DR. PURSER OFFERS
Unlock the deepest look into your nutrient status, antioxidant capacity, and cellular resilience with this upgraded bundle—complete with a personal consultation with Dr. Purser.
This enhanced testing package combines the Cellular Micronutrient Assay (CMA) with the REDOX Panel, adding 50 additional nutrient and antioxidant markers that reveal how well your cells defend, repair, and recover from oxidative stress.
If you want the most complete picture of your metabolic health, this is the test.
WHY THIS TEST IS DIFFERENT
The CMA measures nutrient levels inside your immune cells—not just floating in your bloodstream—giving you a long‑term, accurate view of your metabolic and nutritional status.
The REDOX Panel expands this even further by analyzing 50 additional antioxidant‑related nutrients, helping you understand:
✔️ How well your cells neutralize free radicals
✔️ Your antioxidant reserves and recovery capacity
✔️ How toxins, medications, pollution, and stress impact your cellular health
✔️ Your vulnerability to oxidative damage and inflammation
Together, these tests give you the most complete cellular health assessment available.
WHAT THIS COMPREHENSIVE PANEL REVEALS
✔️ Micronutrient Levels — Vitamins, minerals, amino acids, and essential cofactors
✔️ Antioxidant Status (REDOX) — 50 additional markers tied to cellular defense and repair
✔️ Oxidative Stress Load — How environmental and lifestyle exposures affect your cells
✔️ Metabolic Efficiency — How well your cells produce energy
✔️ Long‑Term Nutrient Patterns — Beyond the “snapshot” of standard serum tests
This is the test for people who want answers—not assumptions.
WHO BENEFITS MOST
Anyone wanting a personalized, data‑driven supplement and nutrition plan
Women navigating fertility, pregnancy, perimenopause, or menopause
Individuals with fatigue, mood swings, low vitality, or chronic stress
High performers, athletes, and those under heavy physical or emotional load
People preparing for or recovering from surgery
Those managing weight, metabolic syndrome, or chronic conditions
Anyone who feels “off” despite normal labs
If you’ve been searching for the root cause of your symptoms, this test brings clarity.
WHAT’S INCLUDED
🩺 30‑Minute Consultation with Dr. Purser A personalized review of your results with expert interpretation and next‑step guidance.
💊 Customized Supplement & Nutrition Plan Tailored recommendations based on your cellular and antioxidant needs, with direct links for easy ordering.
Your kit includes everything needed for your blood draw. Patients are responsible for arranging their own draw with a qualified provider (fees vary). We typically recommend AnyLabTestNow, and you can find a location near you at: www.anylabtestnow.com/locations
Once your sample is collected, contact our office and we’ll schedule your results appointment with Dr. Purser.
THE MOST COMPLETE VIEW OF YOUR CELLULAR HEALTH
Stop guessing. Start measuring. This combined CMA + REDOX panel gives you the data, direction, and expert support you need to transform your health from the inside out.
Order your upgraded bundle today and take control of your long‑term wellness.